FREQUENTLY ASKED QUESTIONS
FRRS1L INFORMATION
What is FRRS1L genetic disorder?
FRRS1L Disorder (pronounced “Frizzle”) is a genetic condition caused by a loss of function mutation in the FRRS1L gene. This gene is responsible for encoding a protein that plays a critical role in ensuring normal development and function of the brain. The loss of function of the FRRS1L gene interferes with proper functioning of the AMPA receptors in the brain, which are essential for neurons in the brain to communicate with each other. In individuals with FRRS1L disorder, disruptions in these neural signaling pathways result in severe lifelong disability.
Children with Frizzle Disorder are born healthy; however, by the age of 2, they experience a severe and sudden regression. The children then go on to develop debilitating epileptic encephalopathy, movement disorders, severe developmental delays, low muscle tone (hypotonia), respiratory failure, and cognitive impairment. They become completely non-mobile and non-verbal, with very limited control of their bodies. This means that they are unable to chew or swallow; they can’t sit, stand or walk; they can’t hold things with their hands; they can’t lift their arms; they can’t turn their head easily from side to side. They are completely dependent on an adult for all movements.
Despite the loss of function and control, FRRS1L disorder children do maintain cognitive awareness, showing their ability to understand and learn. They still present clear emotional responses and communication through sounds and facial expressions. They convey pain, boredom, frustration, happiness, and joy. They can communicate using different speech methods -- some children even use an eye-gaze communication device where they can communicate effectively with their eyes.
Treatment for FRRS1L disorder is ineffective and limited to symptom management and supportive care. Children’s seizures are resistant to epileptic medicines, which means they suffer from ongoing status epilepticus.
Is FRRS1L genetic disorder life limiting and life threatening?
Yes. Due to the rapid and extensive loss of function and abilities, FRRS1L patients’ lives are at risk. Threat to life can include but not be limited to inability to eat/drink/nutrition, airway blockage, respiratory failure, pneumonia, and epilepsy.
What does care for FRRS1L genetic disorder require?
Due to loss of function and abilities, FRRS1L genetic disorder patients require 24/7 care, support, and monitoring. They require support from medical devices such as tube feedings, suctioning, respiratory clearance, and ventilation.
Are there any medications to stop regression or reverse regression; stop/manage epilepsy?
No. There are no effective medications available for FRRS1L disorder; nor treatments which have been shown to prevent regression, improve development or successfully manage seizures in these patients. Patients are highly refractory to seizure medications.
When was FRRS1L discovered as a genetic disease?
FRRS1L genetic disorder was discovered in 2016.
How are patients identified and/or diagnosed with FRRS1L?
Diagnosis of FRRS1L disorder is achieved through genetic testing. To date, basic genetic testing doesn’t include the FRRS1L gene, so whole exome or whole genome sequencing is required for diagnosis. Increased awareness of Frizzle around the world could help doctors include the FRRS1L gene in specific genetic panels in the future.
Is FRRS1L rare?
FRRS1L genetic disorder is found across multiple people groups all around the world. Due to its recent discovery in 2016, and because of limited access to genetic testing, FRRS1L disorder is highly likely to be underdiagnosed and/or misdiagnosed in patients. Most new cases are being found in babies due to early intervention and genetic testing.
Where is FRRS1L found around the world?
FRRS1L has been found around the world, and in multiple people groups. Currently there are children in: Afghanistan, Croatia, Germany, India, Italy, Middle East, Netherlands, Puerto Rico, South Korea, and the USA.
RESEARCH INFORMATION
Is there an upcoming treatment/cure for FRRS1L genetic disorder?
Yes. Gene replacement therapy for FRRS1L disorder is in development. Research has proven that FRRS1L-bred mice which received gene replacement treatment successfully regained function. Doctors and scientists believe this treatment will be life changing for FRRS1L patients.
How does the treatment for FRRS1L work?
FRRS1L patients would receive a one-time dose by lumbar puncture. The treatment uses a virus vector to deliver the normal FRRS1L gene to the brain cells, resulting in functioning AMPA receptors to be built and a return of function to the brain.
When will treatment be available?
The development of gene therapy replacement for FRRS1L disorder is entirely funded by the nonprofit Finding Hope for FRRS1L. The nonprofit raises money to fund and support the development process of this treatment. The timeline for treatment availability for FRRS1L patients depends on how quickly funding goals can be met.
Where are we in the process for treatment development?
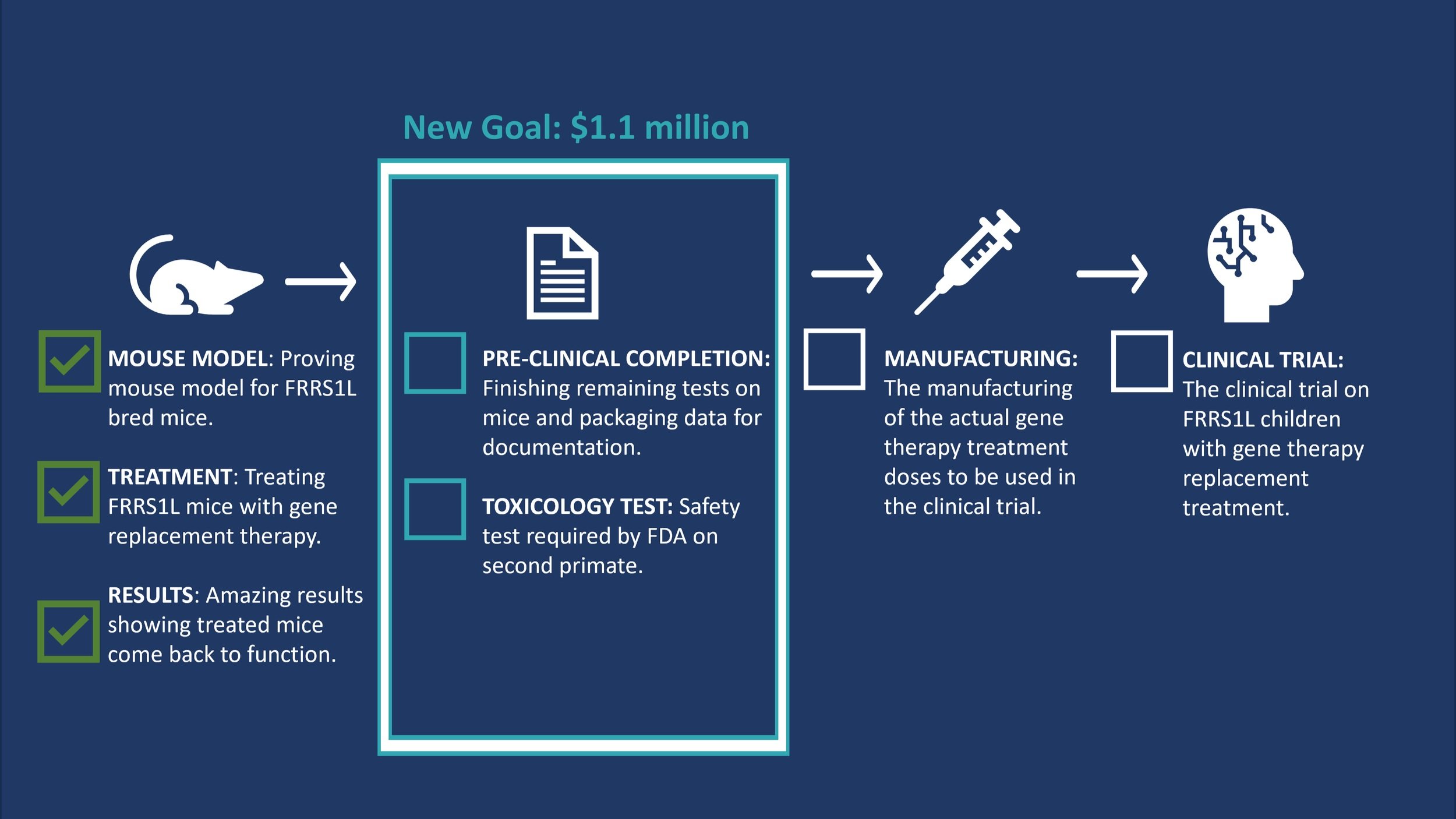
Finding Hope for FRRS1L is working with a clinical partner at the University of Texas Southwestern to develop gene therapy treatment. Currently the team is in the process of finishing the pre-clinical mouse work and preparing an IND package required for the US Food and Drug Administration (FDA) to receive approval to manufacture the drug/treatment to do clinical trials. The next steps in the process include: a Toxicology test (required by the US FDA), manufacturing the drug/treatment, and clinical-trials in FRRS1L disorder patients. See graphic 1.
Graphic 1
FUNDRAISING
What are the fundraising goals for Finding Hope for FRRS1L?
Finding Hope for FRRS1L has a first quick goal to keep the project moving forward of $1.1 million. The $1.1 million would fund the completion of the pre-clinical work, the packaging of information for the US FDA (called the IND package), as well as a toxicology study. The total fundraising goal is $6.1 million. This covers all steps above a well as the manufacturing of the treatment and the clinical trials on children. Learn more about our fundraising plan here. See Graphic 2.
Graphic 2.

